Von Willebrand Disease is a genetic bleeding disorder
|
Mild and more daily symptoms of VWD consist of excessive bleeding in the nose, the mouth (either in the gums or in the incidence of a tooth loss), from the bottom of the foot, and excessive bruising. These can all my indicators of the disease in the process of diagnosis.
When faced with serious situations, such as surgery or a physically traumatic experience, VWD can cause uncontrollable bleeding that can even be fatal. |
Von Willebrand Factor is mutated in VWD
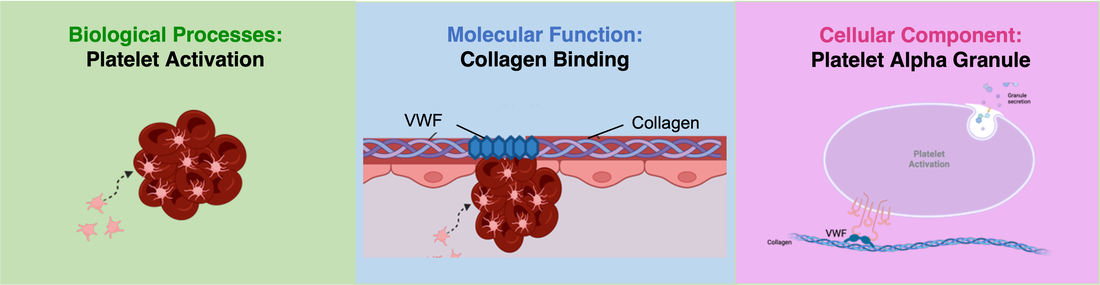
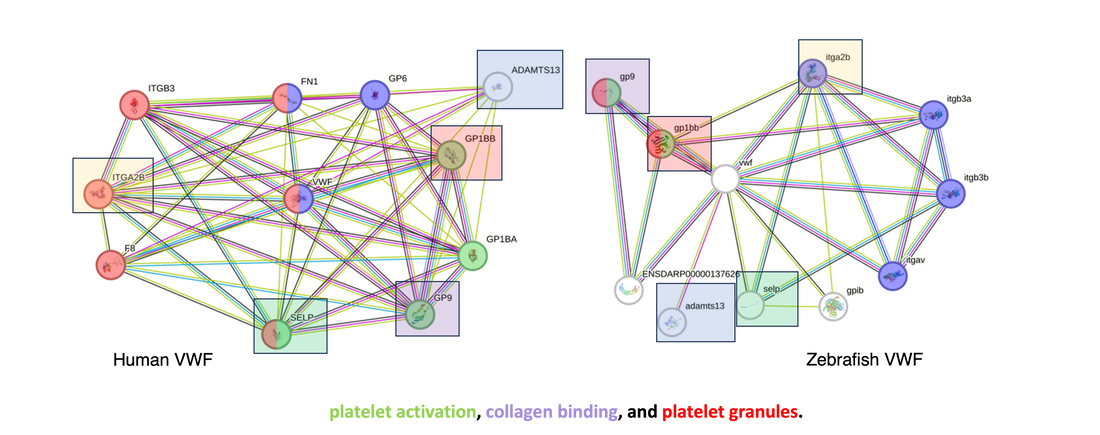
Overall, VWF's gene ontology is represented through its role in platelet activation, collagen binding, and in platelet granules.
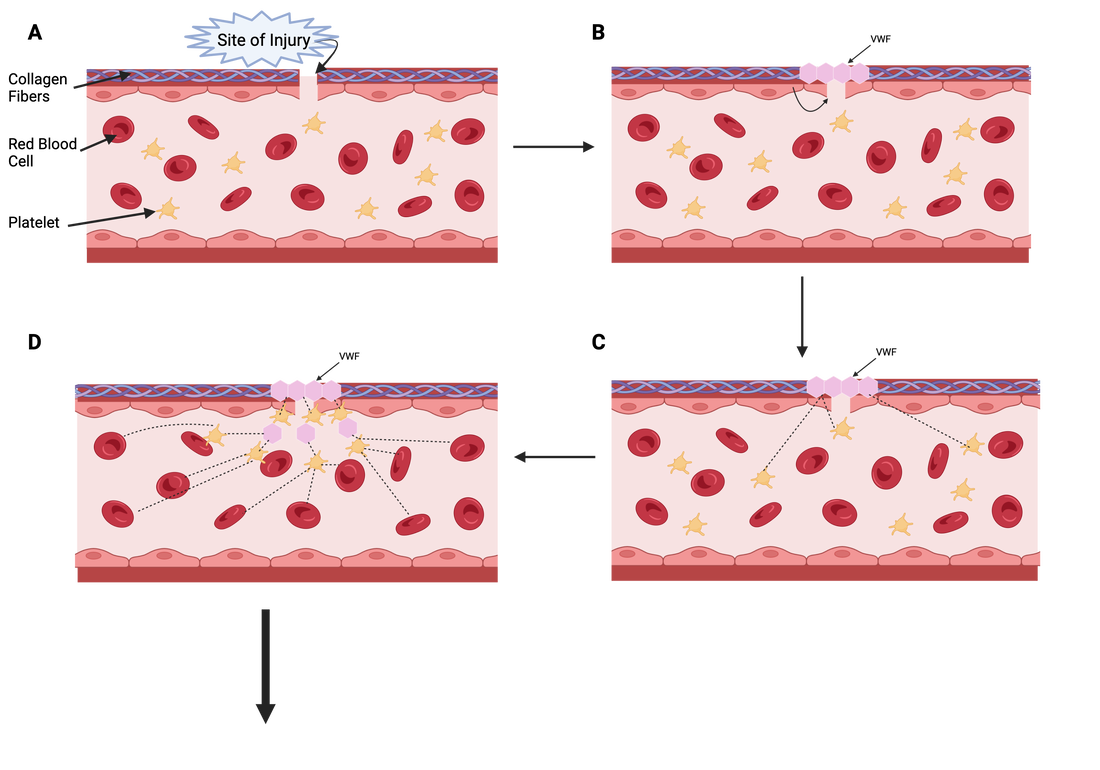
VWF is essential for the formation of blood clots at the site of blood vessel injury. VWF will be released from epithelial cells after vessel injury, and will bind to collagen fibers to bridge the gap at the vessel tear (Figures A and B). The protein can then begin to communicate with coagulation factors circulating in the blood, such as platelets (Figure C). Once the platelets have been activated by the VWF protein, platelet-platelet interactions occur, as well as inclusion of other coagulation factors (Figure D). Together, the VWF-initiated cascade will form a blood clot at the site of injury, effectively preventing excessive bleeding in the body (Figure E).
E
VWF is highly conserved across many model organisms

Phylogeny of VWF model organisms:
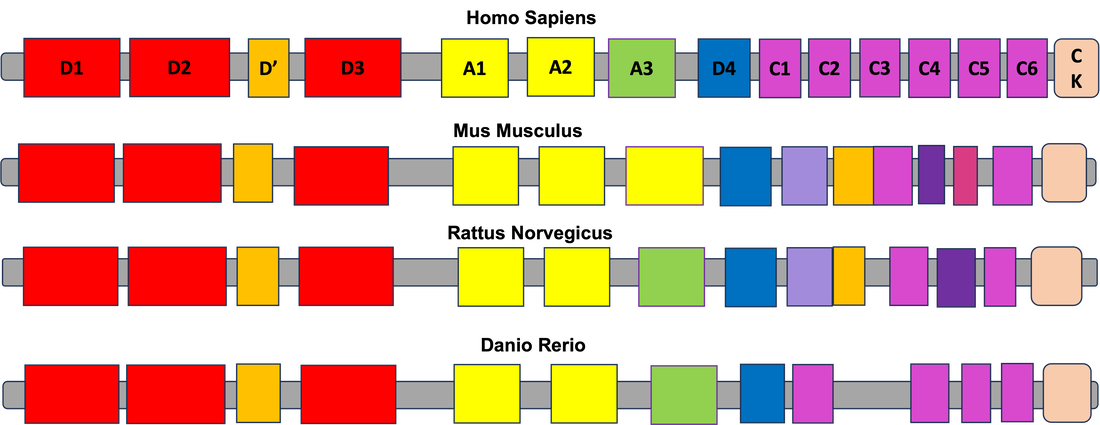
VWF protein sequences are very well conserved across different species, specifically those with circulatory systems. This includes humans, mice, rats, frogs, and zebrafish. The circulatory system controls more than just blood circulation, but also functions in cellular metabolism, organ function, and overall bodily homeostasis. As shown, Danio Rerio (zebrafish) have the most closely conserved VWF sequence in comparison to Homo Sapiens (humans), making them a notable organism for looking at the protein's function.
Further investigation into the protein interactions of the Human VWF as well as the Zebrafish VWF showed that there are significant communications between VWF and other proteins involved platelet activation, collagen binding, and platelet granules. Additionally, some of the same proteins are shared between the two species interaction networks of VWF, which are highlighted above. This data highlights the suggestion that zebrafish may serve as an effective model for VWF research.
Danio Rerio (zebrafish) as a model organism for studying platelets
A protocol that can be used to observe bleeding disorders is known as the laser injury technique. This is when a laser is used to create a tear in the blood vessel wall of a zebrafish, simulating an injury. After doing so, platelets, also known to be thrombocytes in zebrafish, can be fluorescently tagged and their activity can be observed following the laser induced injury. This will allow for the investigation of VWF's role in platelet activation.
Post- Translational Phosphorylation of VWF
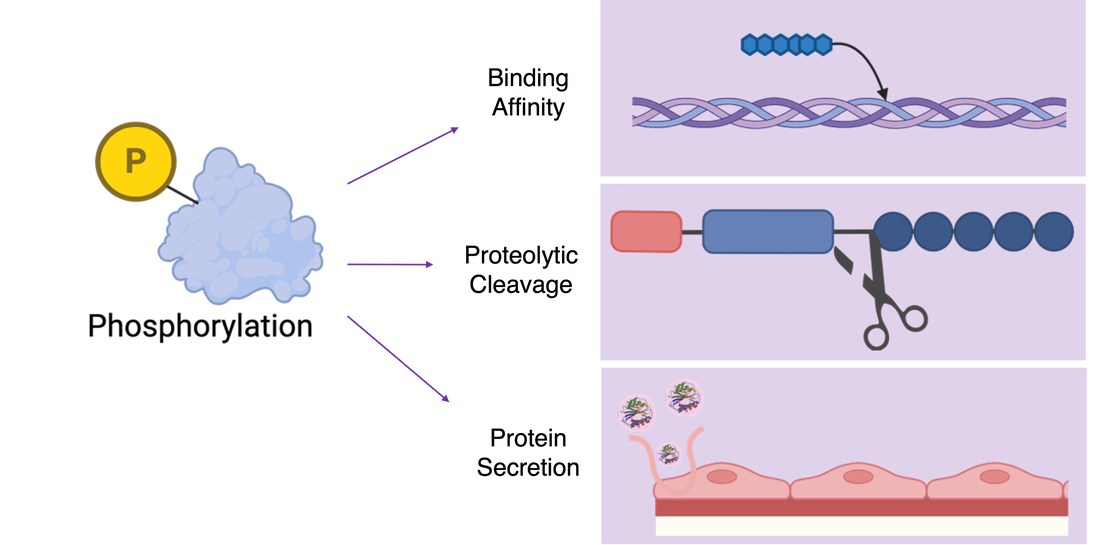
When investigating the role of VWF, it is important to note that the protein itself is modified in many ways, most specifically by phosphorylation. The protein undergoes extensive phosphorylation which plays a major role in the proteins function. The addition of phosphate groups to VWF is known to increase the protein's binding affinity to collagen which starts the coagulation cascade. It is also known that phosphorylation plays a role in the proteolytic cleavage of the protein, which is important since it is such a large protein, and additionally in the secretion of VWF from endothelial cells. These roles suggest that the process of VWF phosphorylation may be pivotal to its functionality.
Gap in Knowledge
|
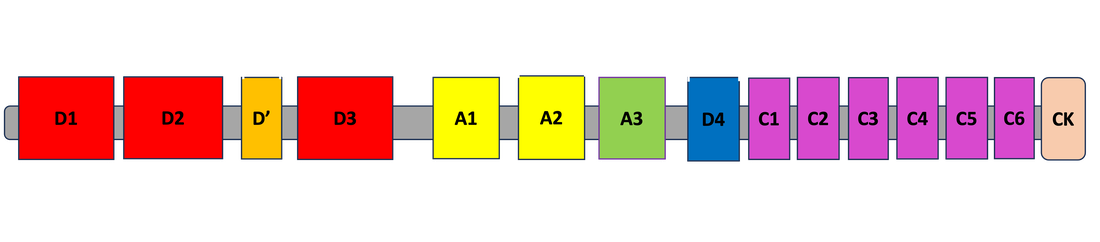
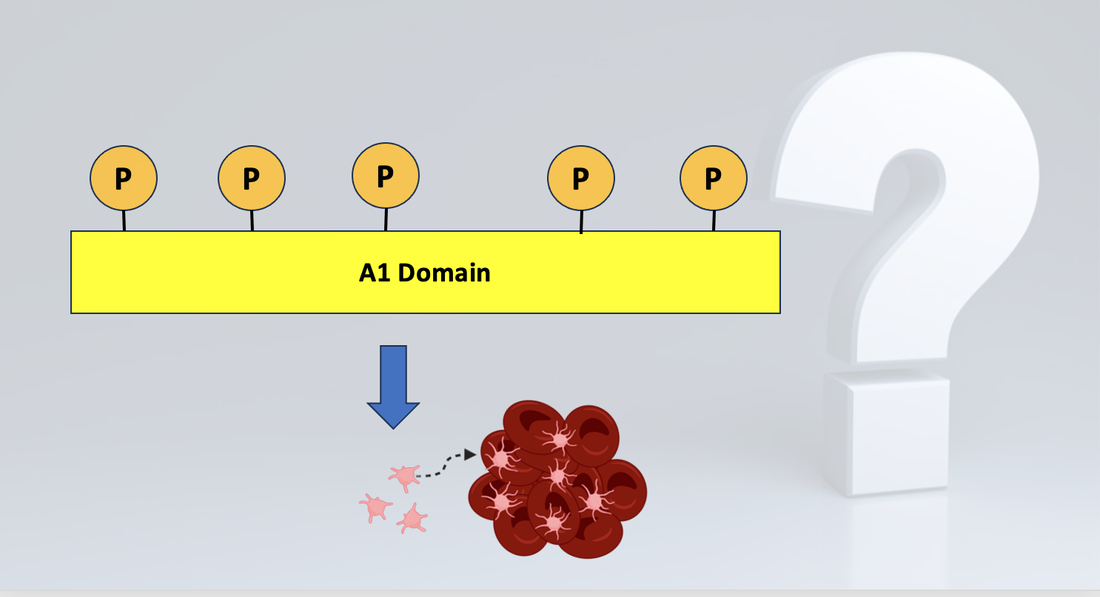
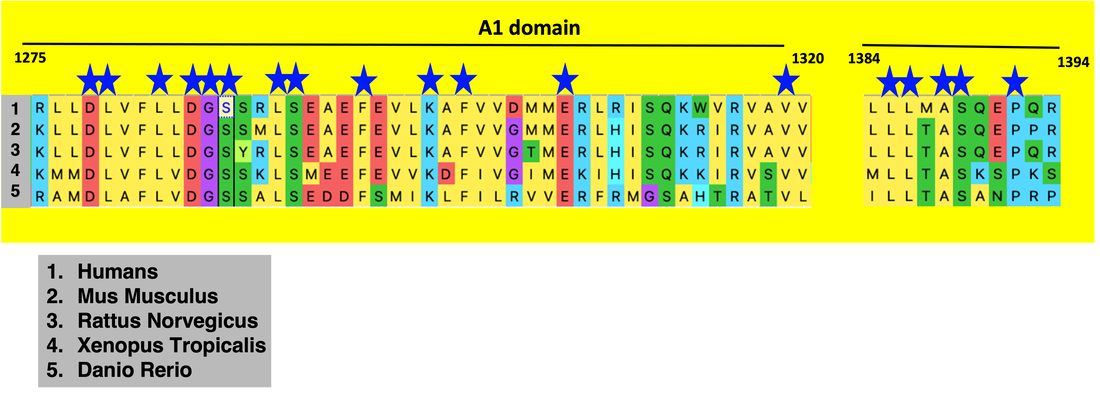
One of the 15 domains of VWF, known as the A1 domain, is recognized as the primary domain responsible for platelet aggregation. While this function is widely accepted, the role and necessity of phosphorylation of the A1 domain is unknown. This project seeks to understand how phosphorylation specifically in the A1 domain effects the VWF protein's ability to activate platelets.
|
Research Goal and Experimental Approach
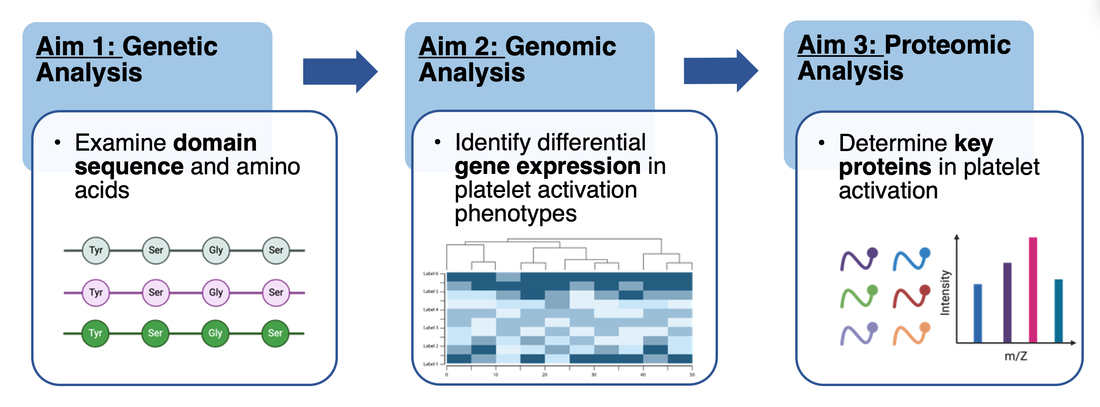
The primary goal of this study is to understand the role of post-translational phosphorylation in the A1 domain on VWF platelet activation. This was researched through 3 different lenses divided into Genetic, Genomic, and Proteomic approaches.
Aim 1: Identify amino acids involved in platelet activation in the A1 domain
|
|
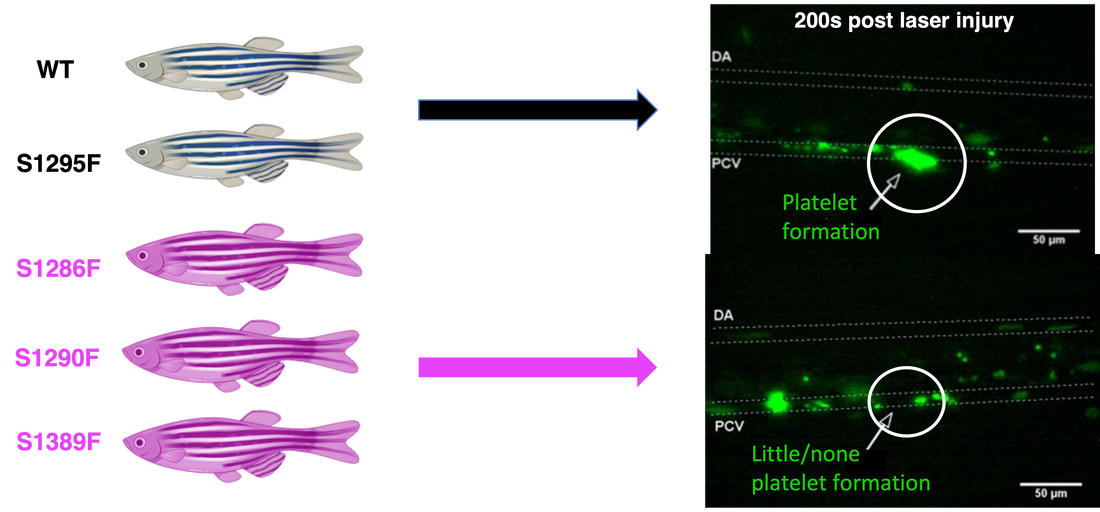
After performing sequence alignment using Clustal Omega, the A1 domain proved to be very highly conserved (blue stars represent conserved amino acid sites). Additionally, I discovered that there were 3 highly conserved and phosphorylated serines in the A1 domain at positions 1286, 1290, and 1389. These serines will be mutated to phenylalanines as they are a very biochemically different amino acid that is not prone to phosphorylation. Additionally, serine at position 1295 will be mutated in a similar fashion to serve as a control, as it is a non-conserved and non-phosphorylated site.
|
Following identification of these amino acid locations, the serines were mutated as mentioned in zebrafish using a CRISPR-Cas9 system. Control group consisted of wild-type and S1295F. Experimental group consisted of the S1285F, S1290F, and S1389F. All of these fish were then examined for their platelet activity following laser-induced injury to the blood vessel. For the control groups, it is expected to see effective platelet activation and fluorescently tagged thrombocytes should be clearly aggregating at the site of injury. However, for the experimental group, I hypothesize that the lack of serine phosphorylation will disrupt the A1 domain's function, and therefore their ability to activate platelets effectively. In this way, the experimental groups should see a lack of thrombocytes aggregating at the site of injury.
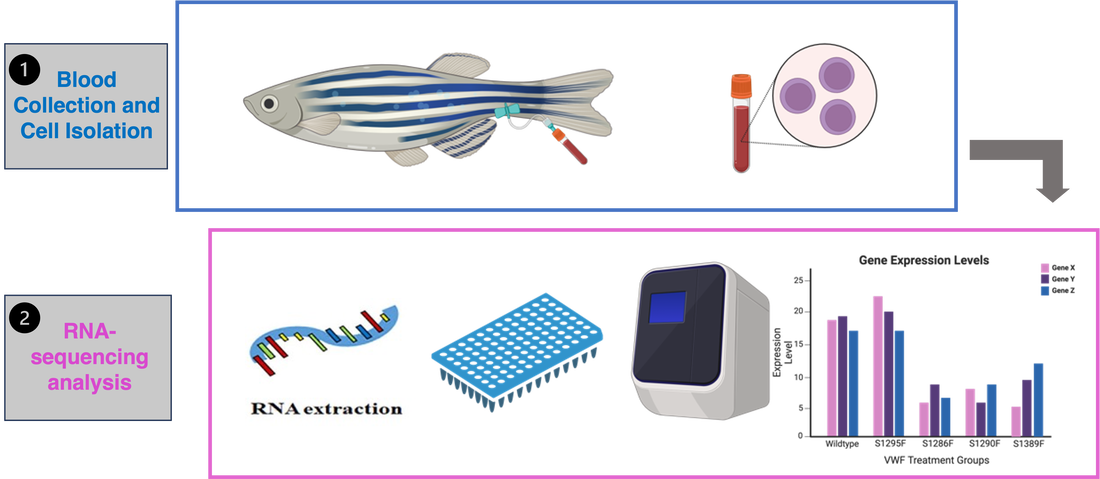
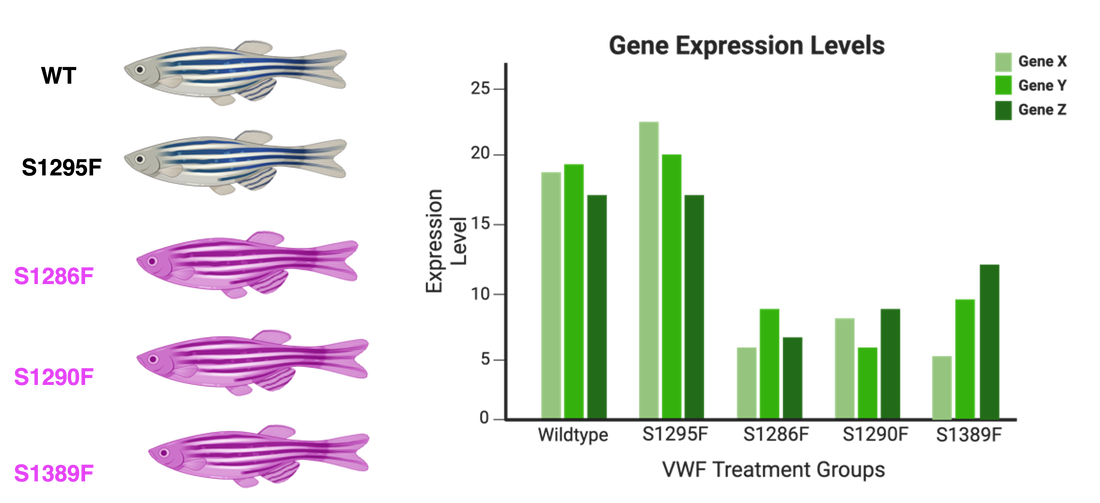
Aim 2: Identify genes involved in platelet activation
|
To examine what genes are involved in this coagulation pathway, blood will be collected from each of the zebrafish in the control and experimental groups, and endothelial cells isolated. Performance of RNA-sequencing and the following analysis will provide information of the genes involved in platelet activation. I hypothesize that the serine mutants in the experimental group will express lower levels of platelet activation genes than the control treatment fish.
|
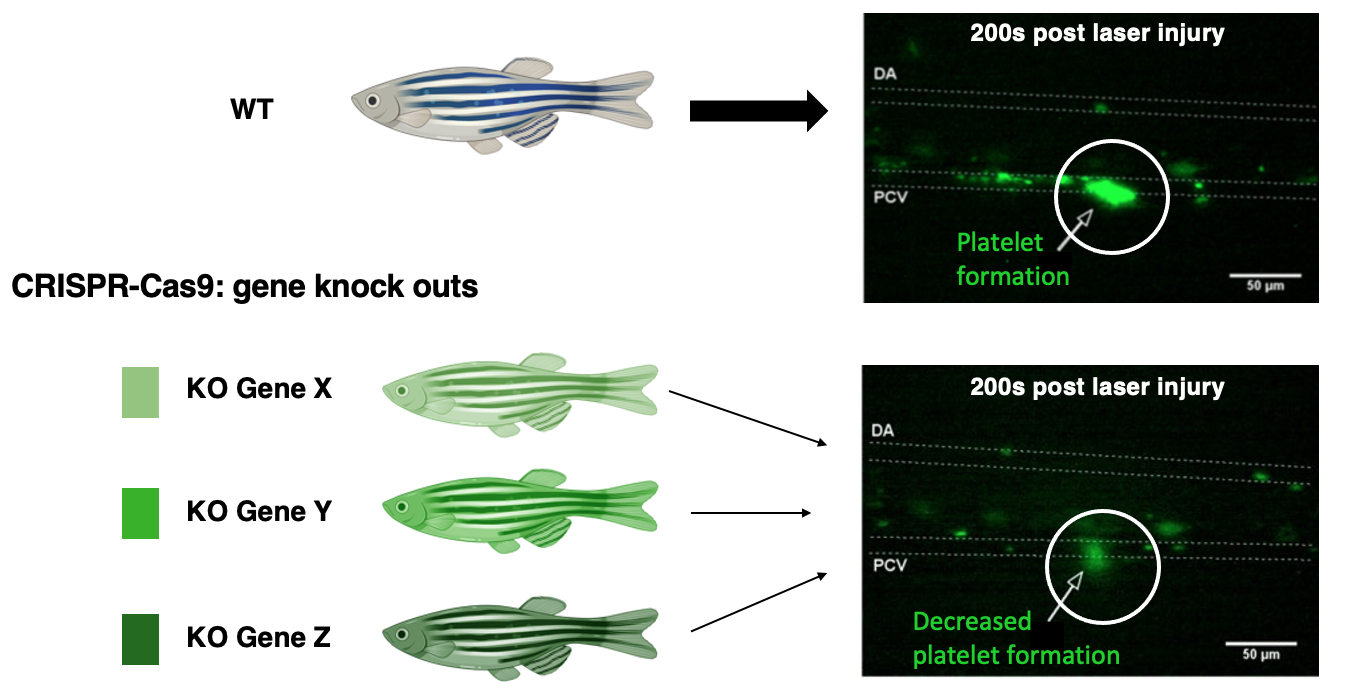
To validate the role of these genes, I will take the genes that are up-regulated in the control groups, in which case platelet activation occurs effectively, and I will knock out those genes in a second line of Wild-Type zebrafish. The new KO zebrafish will be compared to the WT zebrafish; both will undergo laser induced vessel injury, and their platelet activity will be examined. It is expected that the knockout fish will experience some platelet formation, but much less still compared to the WT.
Aim 3: Identify proteins involved in platelet activation
|
|
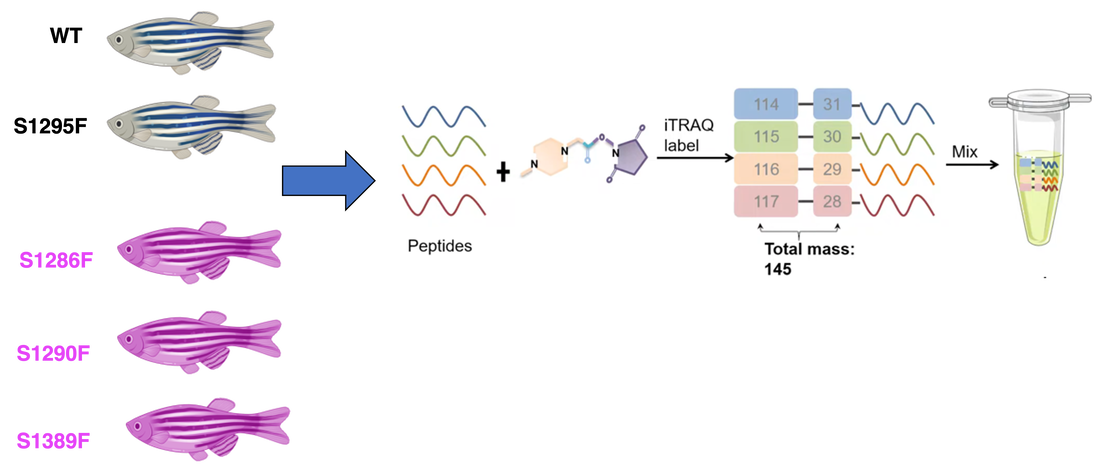
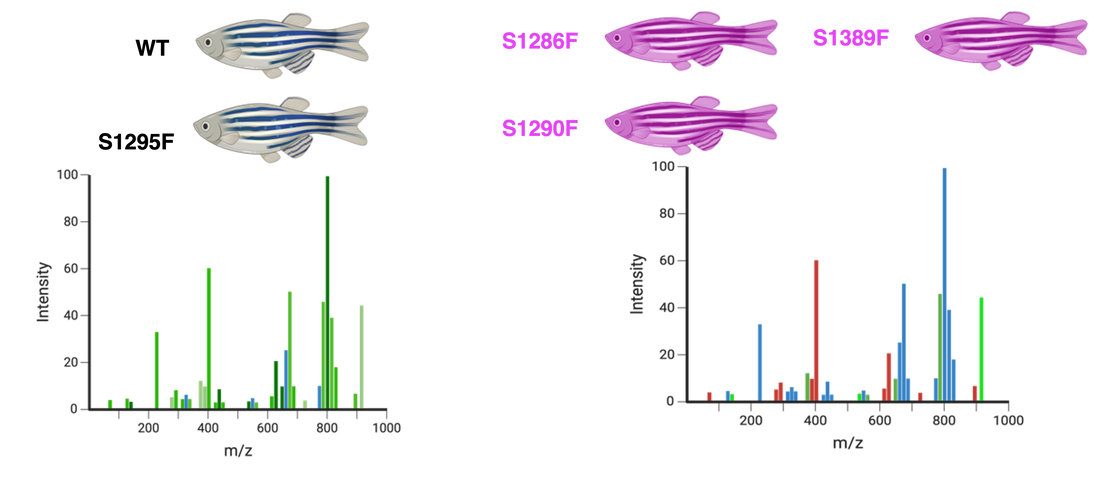
To identify the proteins interacting in the platelet activation process, iTRAQ and Mass Spectrometry will be used. Each of the zebrafish in both the control and experimental groups will be used, and iTRAQ can label the proteins to allow for separate identification. Mass Spectrometry will then be used to analyze the mixture of proteins and identify each. It is expected that experimental serine mutants will show decreased levels of proteins involved in platelet activation.
|
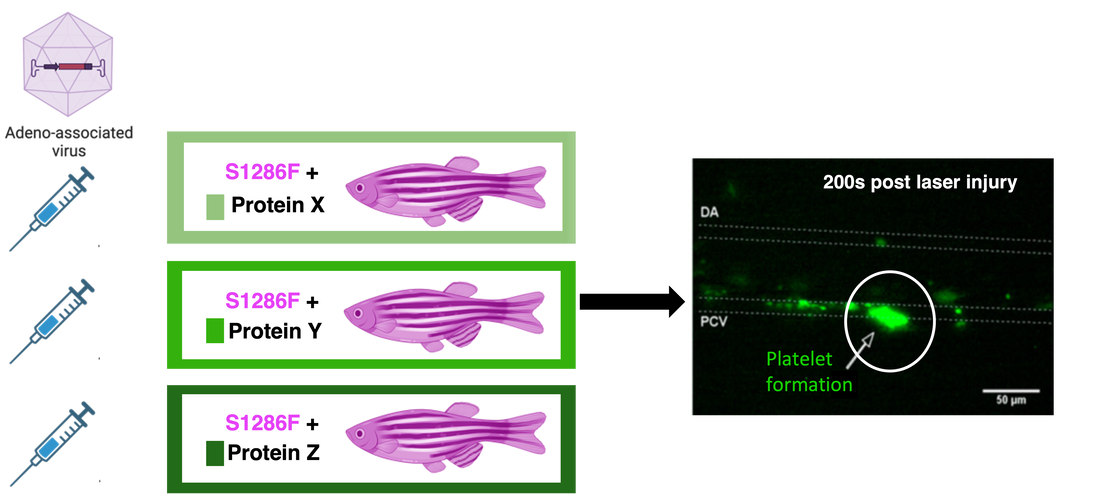
Following Mass Spectrometry, validation of the role of the identified proteins will be done using a CRISPR-Cas9 system. Using solely the S1286F mutant type, proteins identified through Mass Spectrometry involved in platelet activation will be transfected to mutant zebrafish using an adeno-associated virus. This will be done to validate the role of these proteins in platelet activation, and to see if these proteins can rescue the mutant phenotype. I hypothesize that transfection of these target proteins will increase platelet activation of the experimental mutants.
Future Directions
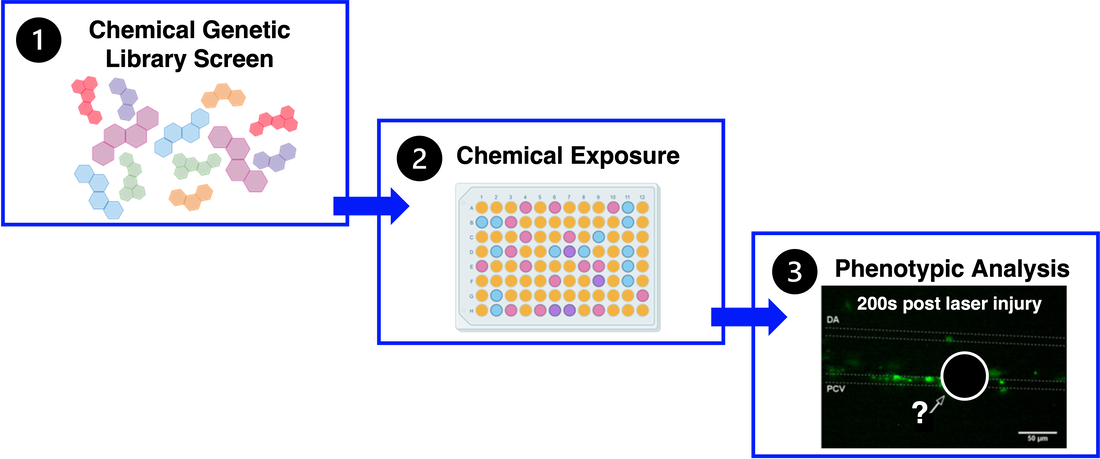
In the future, the results from this research may provide a foundation for looking into chemical genetic approaches to treat VWF dysfunction and VWD. Small chemical molecules discovered from a chemical genetic library screen that are involved in the phosphorylation pathway could be used to continue researching the impact of post-translational phosphorylation of VWF. Similar protocols can be used to assess the phenotypes associated with these molecular studies.
Final Presentation
Final Talk (04/25/2024)
| conte_finaldraft_04.25.24.pdf | |
| File Size: | 4611 kb |
| File Type: | |
|
Final Talk Draft 1 (03/07/2024)
|
Final Talk Draft 2 (03/21/2024)
|
|
| ||||||||||||
References:
[1] Hernández-Zamora, E., Zavala-Hernández, C., Quintana-González, S., & Reyes-Maldonado, E. (2015). Enfermedad de von Willebrand, biología molecular y diagnóstico [Von Willebrand disease. Molecular biology and diagnosis]. Cirugia y cirujanos, 83(3), 255–264. https://doi.org/10.1016/j.circir.2015.05.010
[2] https://www.haemophilia.org.au/bleeding-disorders/von-willebrand-disease/about-vwd/
[3] Biorender
[4] Carrillo, M., Kim, S., Rajpurohit, S. K., Kulkarni, V., & Jagadeeswaran, P. (2010). Zebrafish von Willebrand factor. Blood cells, molecules & diseases, 45(4), 326–333. https://doi.org/10.1016/j.bcmd.2010.10.002
[5] Anne Trafton | MIT News Office. (n.d.-a). New CRISPR-based tool inserts large DNA sequences at desired sites in cells. MIT News | Massachusetts Institute of Technology. https://news.mit.edu/2022/crispr-gene-editing-dna-1124
[6] Sankar, K., Yoon, H. J., Lee, Y. B., & Lee, K. Y. (2022). Evaluation of Reference Genes for Real-Time Quantitative PCR Analysis in Tissues from Bumble Bees (Bombus Terrestris) of Different Lines. International journal of molecular sciences, 23(22), 14371. https://doi.org/10.3390/ijms232214371
[7] Fish, R. J., Freire, C., Di Sanza, C., & Neerman-Arbez, M. (2021). Venous Thrombosis and Thrombocyte Activity in Zebrafish Models of Quantitative and Qualitative Fibrinogen Disorders. International journal of molecular sciences, 22(2), 655. https://doi.org/10.3390/ijms22020655