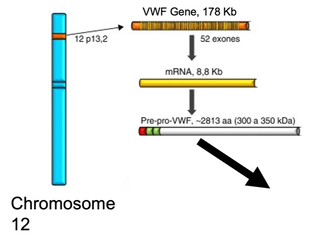
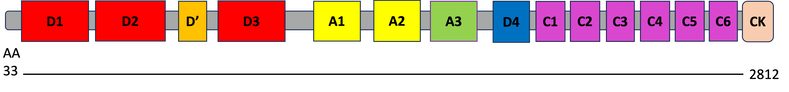
VWF Domains
Protein Domains
Protein Domains are locations in a gene that have specific functions and specialized structural components. Typically, domains have lower level functions that contribute to the protein's function as a whole in the body. These domains can be involved in protein-protein interactions separate from the entire protein's main function, and can provide insight into how a protein may be mechanistically playing a role in various biological and molecular pathways. It is possible to find similar domains in other proteins [1].
The VWF gene has 15 different protein domains that are relatively well-conserved across model organisms. (currently am missing xenopus tropicalis below because I need to re-do it)
The VWF gene has 15 different protein domains that are relatively well-conserved across model organisms. (currently am missing xenopus tropicalis below because I need to re-do it)
Discussion
The VWF protein is a long peptide made up of 15 different functional domains. Of interest for this project is the A1 domain, which is shown to be one of the most highly conserved domains across all homologs with minimal variation in amino acid position. The A1 domain is associated with platelet activation, although the mechanisms by which it functions to do so are still incompletely understood [2]. It is known, however, that post-translational phosphorylation of the VWF protein is associated with increased affinity for platelets to the blood vessel endothelium following platelet aggregation by VWF protein [3]. Observations from these results in protein domain research serve as the foundation of this projects' interest in the A1 domain and it role in platelet activation, using the highly conserved domain in Zebrafish (Danio rerio).
References:
[1] Embl-Ebi. (n.d.-b). What are protein domains?. What are protein domains? | Protein classification. https://www.ebi.ac.uk/training/online/courses/protein-classification-intro-ebi-resources/protein-classification/what-are-protein-domains/
[2] Goodeve A. C. (2010). The genetic basis of von Willebrand disease. Blood reviews, 24(3), 123–134. https://doi.org/10.1016/j.blre.2010.03.003
[3] Karlaftis, V., Perera, S., Monagle, P., & Ignjatovic, V. (2016). Importance of post-translational modifications on the function of key haemostatic proteins. Blood coagulation & fibrinolysis : an international journal in haemostasis and thrombosis, 27(1), 1–4. https://doi.org/10.1097/MBC.0000000000000301
[1] Embl-Ebi. (n.d.-b). What are protein domains?. What are protein domains? | Protein classification. https://www.ebi.ac.uk/training/online/courses/protein-classification-intro-ebi-resources/protein-classification/what-are-protein-domains/
[2] Goodeve A. C. (2010). The genetic basis of von Willebrand disease. Blood reviews, 24(3), 123–134. https://doi.org/10.1016/j.blre.2010.03.003
[3] Karlaftis, V., Perera, S., Monagle, P., & Ignjatovic, V. (2016). Importance of post-translational modifications on the function of key haemostatic proteins. Blood coagulation & fibrinolysis : an international journal in haemostasis and thrombosis, 27(1), 1–4. https://doi.org/10.1097/MBC.0000000000000301
This web page was produced as an assignment for Genetics 564, a capstone course at UW-Madison.
Michelle Conte | [email protected] | Last edited 05/08/2024 | www.genetics564.weebly.com